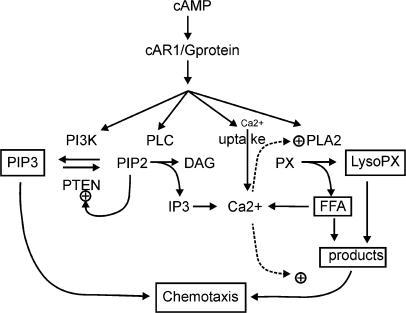
PIP3 is the effector of multiple downstream targets of thephosphoinositide 3 kinase (PI3K) pathway. Forced expression of wild-type but not mutant tumor suppressor PTEN in CEF decreased survivin mRNA levels. proteins involved in phospholipid metabolism (Table1).These phosphoinositides direct two major independent signaling cascades. Hence, the PTEN-opathies serve as a prototype for bedside to bench, and back to the bedside, practice of evidence-based precision medicine. To investigate the role of PI3K pathway in the regulation of survivin, we expressed an active form of PI3K, v-P3k in chicken embryonic fibroblast cells (CEF), and found that overexpression of PI3K-induced survivin mRNA expression. 1 AKT can have a number of downstream effects such as. PI3K activation phosphorylates and activates AKT, localizing it in the plasma membrane. Therefore, it is directly related to cellular quiescence, proliferation, cancer, and longevity. Such insights not only teach us about biological mechanisms in states of health and disease, but also enable more accurate gene-informed cancer risk assessment, medical management, and targeted therapeutics. The PI3K/AKT/mTOR pathway is an intracellular signaling pathway important in regulating the cell cycle. The ber of PI3K-PIP important 3 pathway downstream influences kinase the activity enzymes, of such a numas HN Binimetinib Idelalisib. Germline and somatic mosaic mutations in genes encoding components of the PI3K/AKT/mTOR pathway downstream of PTEN predispose to syndromes with partially overlapping clinical features, termed the “PTEN-opathies.” Experimental models of PTEN pathway disruption uncover the molecular and cellular processes influencing clinical phenotypic manifestations. Germline pathogenic PTEN mutations cause PTEN hamartoma tumor syndrome (PHTS), featuring various benign and malignant tumors, as well as neurodevelopmental disorders such as autism spectrum disorder. We conclude that IVIG treatment represents a promising therapeutic approach potentially able to decrease mortality and morbidity after ICH in experimental models. The tumor suppressor phosphatase and tensin homolog (PTEN) classically counteracts the PI3K/AKT/mTOR signaling cascade. Furthermore using siRNA-induced in vivo knockdown approach we demonstrated that beneficial effects of IVIG were mediated, at least partly, via SHIP1/PIP3 pathway.
